Electrocardiogram, Stress Reduction, Vagus Nerve Stimulation.
Abstract: Non-invasive vagus nerve stimulation is quickly gaining popularity as an alternate treatment for a variety of health conditions. In view of contradictory findings from earlier studies, the authors explored the effects of cervical vagus nerve stimulation for stimulating the parasympathetic nervous system to relieve stress. The cervical region was stimulated and ECG data were collected during pre- and post-stimulation trials to study changes in the low-frequency (LF) and high-frequency (HF) heart rate variability parameters (HRV). Comparing pre- and post-stimulation readings for five patients reveals a rise in the HF component and a decrease in the LF component, indicating that VNS generated increased parasympathetic activity. After stimulation, the LF/HF ratio decreased for all subjects, with an average drop of 64.5% seen. Overall, this study demonstrates the viability of employing the cervical region as a stimulation site to stimulate the vagus nerve, with the cervical region being easier to administer than many alternative stimulation locations, while still being beneficial for stress reduction.
1. INTRODUCTION
Constant daily demands are a cause of increased stress and worry, which can lead to a variety of mental diseases. Stress is the biggest contributory factor and primary cause of diseases such as cardiovascular, chronic skin issues, chronic cluster headaches, and other psychiatric disorders. As the typical success rate of most pharmaceuticals is less than fifty percent and they can induce unbearable unfavorable side effects, treating patients with stress-related disease using drug-administered techniques is not a viable treatment option for physicians. In recent years, alternative treatments have been developed to address these concerns and reduce the severe side effects of medications. In particular, approaches based on nerve stimulation are being extensively investigated. Due to their capacity to modulate the neurological system, these techniques are frequently called neuromodulation. Vagus nerve stimulation (VNS) has been extensively studied since 1990 utilizing implanted or invasive VNS devices, among other neuromodulation methods. The vagus nerve is the tenth cranial nerve and is composed of around 80% afferent fibers reaching into the brain and 20% efferent fibers extending to the remainder of the body. It is the principal parasympathetic innervation of the autonomic nervous systemSince the first human implant of VNS devices in 1989, over one hundred thousand patients worldwide have been treated with VNS, and the vagus nerve is commonly regarded as protective, defensive, and soothing. VNS was recently licensed for therapeutic use in the United States for patients over the age of 12, as well as those with drug-resistant epilepsy and depression. Incongruent results have been reported in the scientific literature, with one study finding no difference between sham stimulation and VNS. In this work, we evaluated the effects of vagus nerve stimulation (VNS) at the cervical site and its stress-reduction impact. As a promising indicator of stress, Heart Rate Variability (HRV) was utilized in this study. HRV is the variation in the time between successive heartbeats. It is sensitive to changes in the sympathetic and parasympathetic nervous systems, which can be used to infer stress levels. According to a number of studies, when stress is created, the HRV variable HF lowers and LF increases to reduce parasympathetic activity. Recent research has focused on non-invasive ways of VNS, which can sidestep complex implantation procedures and lessen hazards such as infection. To address this, electrical stimulation of the cervical vagus nerve has been studied utilizing bioelectronics, with an emphasis on its therapeutic effects. Stimulating the auricular branch of the vagus nerve (ABVN), also known as Alderman’s nerve or Arnold’s nerve, has shown efficacy in treating depression. To provide electrical stimulation to the auricular branch, either the cymba conchae or tragus may be used, as these locations contain the majority of vagus nerve fibers. Despite the fact that the conchae contains 100 percent vagus fibers, it is easier to deliver electrical stimulation to both walls of the ear using a proper stimulation clip on the tragus. For any parasympathetic response to be activated, the frequency and pulse width of electrical stimulation are critical characteristics that must be selected with care. The stimulus pattern influences whether parasympathetic or sympathetic responses are activated. To stimulate the parasympathetic system, stimulation frequencies were developed which typically trigger the sympathetic response. The frequency selectivity of the epidermal barrier between electrodes and nerve cells is a problematic aspect.
In experimental research involving auricular VNS, a stimulation frequency of 25 Hz is typically employed, and a pulse width of 200 s is regarded as effective and safe for prolonged stimulation. While it is uncommon to stimulate the vagus nerve at the cervical level, there is a great deal of potential at the cervical level, where the vagus nerve is particularly amenable.
2. METHODOLOGY
A stimulation regimen as depicted in Figure 1 was used to provide VNS at the cervical level. Three trials were conducted over two sessions, beginning with a five-minute rest time for the baseline recording, a six-minute stimulation/placebo session, and a five-minute post-stimulation session. As described in the section titled “Hardware Design,” specialized hardware was created for administering the stimulation.
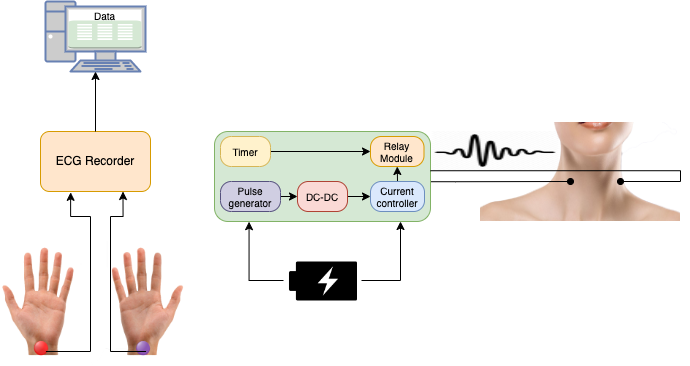
2.1 Hardware design
Figure 2 shows the different parts of the hardware that make it work. To get the ECG data, a Biosemi Active Two system with a separate battery pack was used. Conductive gel was used to attach two electrodes to each participant’s left and right wrists. The reference and ground were the CMS and DRL electrodes. (Pulsetto) We used an Arduino microcontroller, which was programmed to make PWM pulses for our protocol, to make the stimulation pulse. Along with the hardware prototype, code was made to make a signal with a custom pulse width, frequency, and amplitude. This was sent to a DC-DC controller, which controlled the output current, which was limited to 2 mA for safety and had pulse amplitudes of 2.2 volts. When the stimulator was turned on, another Arduino microcontroller was used to set the stimulation time to 6 minutes and to control a dual pole, dual throw relay module that turned the pulses on and off. Figure 3 shows how the output of the relay module was connected to two small, custom-made electrodes that can be attached to both sides of the neck. Based on other VNS experimental studies, we chose the pulse width and rate, but in this case, we stimulated the cervical site. The hardware prototype is a stand-alone unit with a frequency of stimulation that has already been set. It is powered by a 5 V DC battery pack, which makes it safer to use and keeps it from being affected by power surges and interference from outside sources.
2.2 Procedure
For this study, 15 healthy people (seven women and eight men, with an average age of 34.6 ± 12.5 years) took part in two sessions where they were given stimulation and a fake treatment on different days. For ECG collection, each participant sat in a comfortable chair and had electrodes attached to both wrists with gel. The current stimulator had conductive paste on it and was hooked up to the left and right sides of the cervical spine. In each session, three separate ECG recordings were made, starting with a 5-minute baseline (pre-stimulation), then a 6-minute session without stimulation (placebo), and ending with a 5-minute “post-stimulation” recording, as shown in Figure 1. The same person was recorded again on a different day, once with a placebo and once with an active current. Participants didn’t know which session was a placebo and which one had active VNS, so the order of stimulation and placebo was chosen at random. The collected data were made anonymous and then looked at to study how current stimulation affects the brain.
2.3 Data Processing
For each participant, two different sets of data were looked at for the placebo and VNS sessions. The EEGLAB plug-in for MATLAB was used to change the 128 Hz sample rate of the 24 bit Biosemi Data Format (BDF) recordings to the European Data Format (EDF). Data from 1 to 35 Hz was filtered with an IIR Elliptic filter, and the ECG was taken from the channel for the left wrist. The channel for the right wrist was used as a reference. R-R peaks were found using a peak detection method in MATLAB that involved a predefined threshold that was checked by hand for each experiment to make sure that artifact-induced peaks were left out. These R-R intervals were put into the Kubios HRV 3.3.1 analysis software to find the values for LF (0.04–0.15 Hz), HF (0.15–0.4 Hz), and the LF/HF ratio.
3. RESULTS AND DISCUSSION
The LF and HF components of HRV can be estimated to assess the impact of VNS. Based on these findings, it is clear that VNS lowered LF values while increasing HF values, indicating that the parasympathetic nervous system was more activated than the sympathetic nervous system. The overall LF/HF ratio reduction from the pre-VNS and post-VNS sessions was also calculated. An average reduction of 64.5 percent was seen across all fifteen participants between pre- and post-VNS sessions. Meanwhile, a 6.8 percent drop was seen between pre- and post- placebo sessions. Figure 4 also depicts the decrease in electrocardiogram-derived respiration (EDR).
When someone sits down calm for short periods of time, a little reduction is to be expected; nevertheless, the larger reduction found for the VNS sessions may suggest the efficiency of the stimulation in lowering stress. Figure 4 depicts an example of an HRV frequency analysis from one participant for both pre- and post-VNS sessions, with the higher HF rise over LF increase in post-VNS indicating an increase in parasympathetic activity vs sympathetic activity. Changes in HRV parameters as a result of VNS have also been documented in other investigations with comparable frequency.
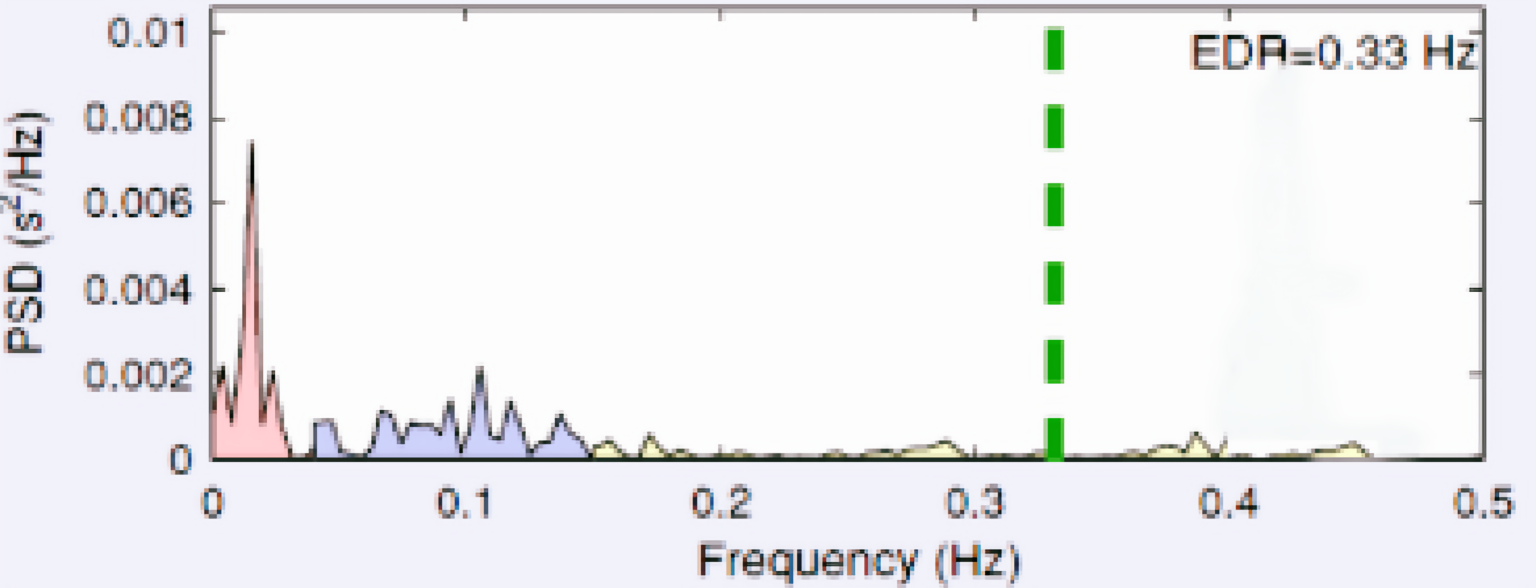
Pre VNS session from one participant
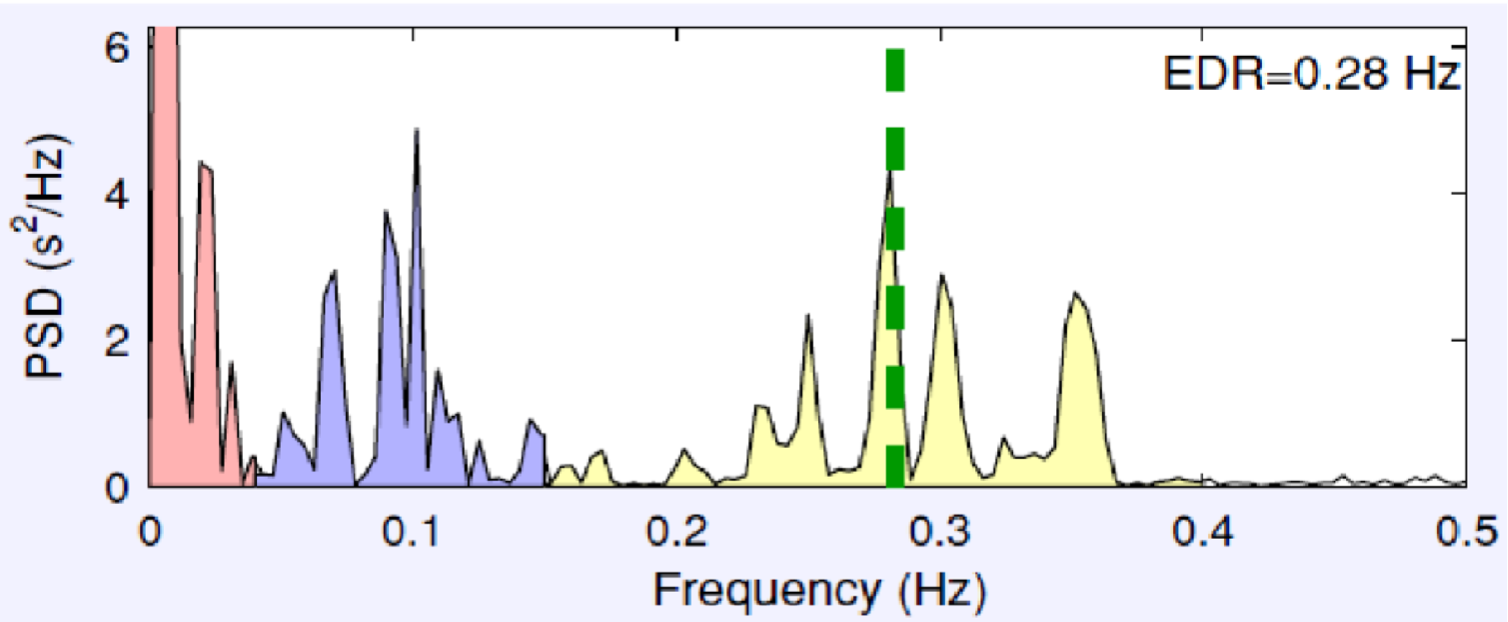
Post VNS session from one participant
Figure 4: Pre – and Post – VNS session from one participant.
4. CONCLUSION
In this study, the possibilities of non-invasive stimulation of the vagus nerve in the cervical site. In the experiments, the left and right cervical sites were stimulated for 6 minutes to bring on a parasympathetic response and make people feel less stressed. Using portable hardware and software (Pulsetto) that can run on its own, and giving it during stimulation. Using electrodes that were made just for the neck, the vagus nerve was easily stimulated. Even though the stimulation only lasted for 6 minutes, all of the participants saw a decrease in the LF/HF ratio after VNS. This was because the LF components went down and the HF components went up, which showed that the parasympathetic system was more active than the sympathetic system. For this study, the VNS period was limited to 6 minutes so that the effects of non-invasive stimulation could be studied. However, future studies could look at longer stimulation times or the effects of regular sessions with clinical patients.









